
















Images for illustrative purposes only
Important BOTOX® Safety Information
BOTOX® Cosmetic (onabotulinumtoxinA) Important Information Indications
BOTOX® Cosmetic (onabotulinumtoxinA) is indicated in adult patients for the temporary improvement in the appearance of:
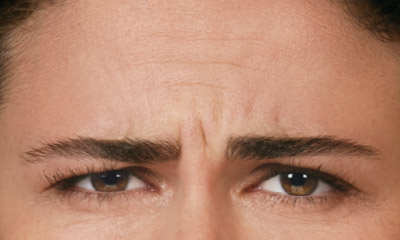
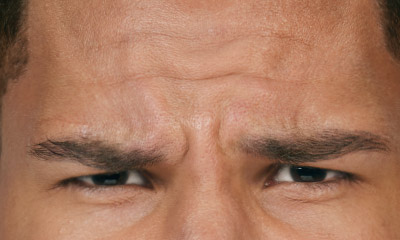
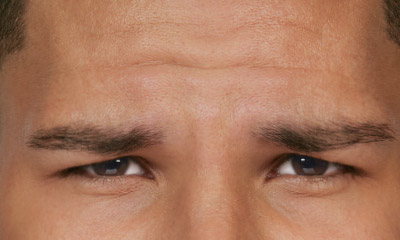
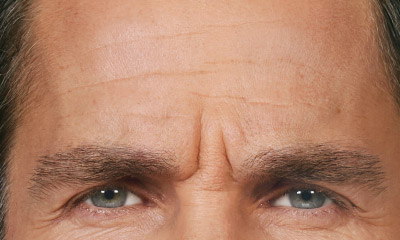
- Moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity
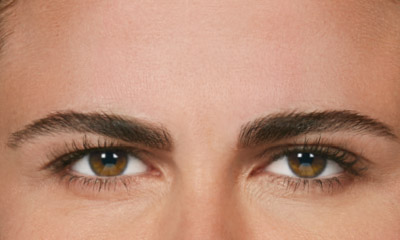
- Moderate to severe lateral canthal lines associated with orbicularis oculi activity
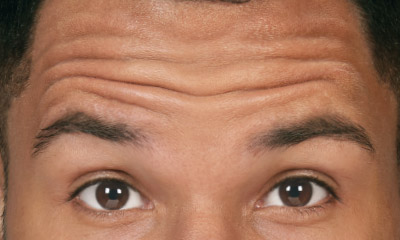
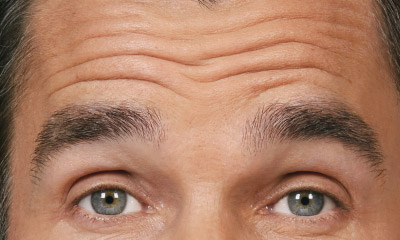
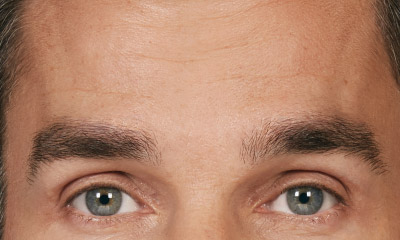
- Moderate to severe forehead lines associated with frontalis activity
Important Safety Information, Including Boxed Warning: Distant Spread Of Toxin Effect
Postmarketing reports indicate that the effects of BOTOX® Cosmetic and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence, and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening, and there have been reports of death.
Important Juvederm Safety Information
JUVÉDERM® Collection of Fillers Important Information
Indications
JUVÉDERM VOLUMA® XC injectable gel is indicated for deep (subcutaneous and/or supraperiosteal) injection for cheek augmentation to correct age-related volume deficit in the mid-face in adults over the age of 21.
JUVÉDERM® Ultra XC and JUVÉDERM® Ultra Plus XC injectable gels are indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds).
JUVÉDERM VOLLURE™ XC injectable gel is indicated for injection into the mid-to-deep dermis for correction of moderate to severe facial wrinkles and folds (such as nasolabial folds) in adults over the age of 21.
JUVÉDERM® Ultra XC injectable gel is indicated for injection into the lips and perioral area for lip augmentation in adults over the age of 21.
JUVÉDERM VOLBELLA® XC injectable gel is indicated for injection into the lips for lip augmentation and for correction of perioral rhytids in adults over the age of 21.

